
The first atomic bomb test at the Trinity Site in south-central New Mexico fused the surrounding sand into a green glassy substance dubbed "Trinitite". Trinitite contains fallout products from the explosion plus small amounts of Uranium and Plutonium from the bomb. A sample of Trinitite was purchased from a mineral dealer and a spectrum obtained to see if the radioactive materials inside could be indentified. The image below shows the specimen tested.

The gamma-ray spectrum for the Trinitite is shown below. Four 0.093" thick Plexiglass sheets were used between the Trinitite and the probe to block the beta particle that are also being emitted. These beta particles otherwise produce a background continuum that swamps most of the peaks visible in the spectrum. The effects of background radiation have been subtracted from this plot.
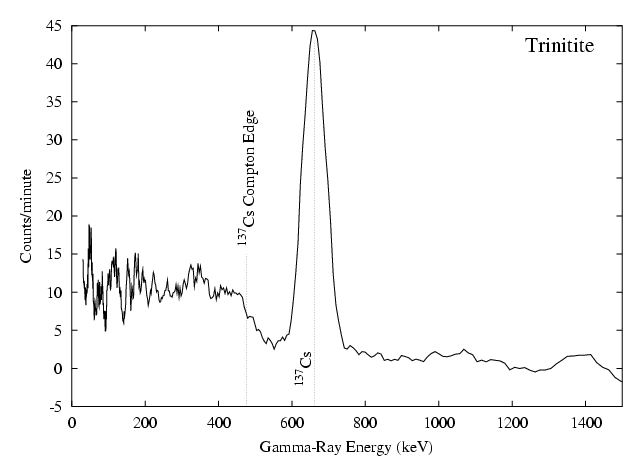
The only radioactive isotope that can be identified from the spectrum is Cesium 137, a typical atomic bomb fallout product. This can be identified from the single peak at 662 keV. The Compton edge for this energy is also visible. The value shown corresponds to the theoretical maximum Compton recoil electron energy lying 184 keV below the photopeak. The Cs-137 peak is the only part of the spectrum that significantly exceeds the background levels. The corresponding Compton continuum covers an energy region where the background readings are relatively high thus the signal-to-noise ratio is low leading to the noisy data seen here.
| Return to Home Page | Latest update: July 18, 2004 |